 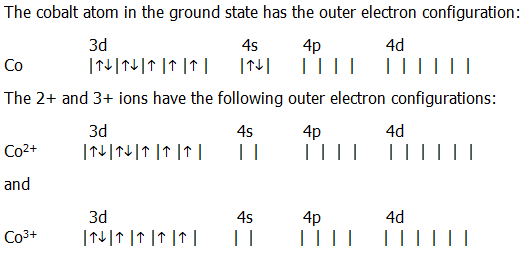
The electronic configuration of cobalt will consist of filling 27 electrons according to the Aufbau principle. We have been given cobalt which has atomic number z = 27. Electron configuration The arrangements of electrons above the last (closed shell) noble gas. The s, p, d, f subshells are written along with the number of the shell, like 1,2, 3, etc. Atomic number The number of protons in an atom. The filling of electrons in various orbits is according to a principle of Aufbau that takes place from the lower energy level to the higher energy level. Electronic configuration of any element consists of filling the orbital or the s, p, d, f sub – shells with electrons. The Electronic configuration of any atom tells us the total number of electrons which is equal to the total number protons in that atom that is equal to the atomic number of that element. The electronic configuration requires filling of orbital or sub shells (s, p, d, f) according to Aufbau principle. Alternatively, you can count the superscripts of the energy levels until you get to ten. As you can see, sodium has a 3s 1 that neon does not have, therefore, the noble gas configuration for sodium would be Ne3s 1. It is identified through atomic number, as the atomic number of an atom tells us the total number of electrons in that atom. The full electron configuration for sodium is 1s 2s 2 2 2p 6 3s 1 and neon is 1s 2s 2 2 2p 6. Phenom., 1980, 21, 275.Hint: The electronic configuration of any element tells us the total number of electrons present in that atom. Hello Guys,Determining the electron configuration of any element is an easy and quick process, given that you know all the required information and general c. In a D4h site with a strong elongation the 3A2g term of cobalt (+III) can be stabilized with an electronic configuration intermediate between low spin. The ground state electron configuration of ground state gaseous neutral cobalt is Ar.3d7. What is the short-hand electron configuration of cobalt(II), Co²+ ion Enter the electron configuration using no spaces or superscripts using the proper case letters (e.g., He 2s² 2p4 would be entered as He2s22p4). Mårtensson, "Core-Level Binding Energies in Metals," J. Cobalt atoms have 27 electrons and the shell structure is 2.8.15.2. Write the electron structure of the two cations. Cobalt forms cations in two oxidation states, Co 2+ and Co 3+. An investigation showed the cause to be the absence of sufficient cobalt in the soil. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. In one area of Australia, the cattle did not thrive despite the presence of suitable forage. The ground state electron configuration of cobalt is 'Ar3d'7'4s'2. In a neutral cobalt atom, there is also 27 electrons. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Cobalt has an atomic number of 27, which means that its atoms have 27 protons in their nuclei. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The data are adapted from references 1-3. English: An SVG periodic table of the elements, which includes name, atomic mass, electron configuration, first ionization energy, and electronegativity. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. Science Chemistry Chemistry questions and answers Write the full electron configurations of cobalt metal, Co, and one of its ions, Co3+. Nickel has the atomic number 28 so it has the electron configuration 1s22s22p63s23p64s23d8. All values of electron binding energies are given in eV. This page shows the electron configurations of the neutral gaseous atoms in their ground states. 1967, 47, 1300.Įlectron binding energies Electron binding energies for cobalt. 
These effective nuclear charges, Z eff, are adapted from the following references: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
